Smoking Induces the Accumulation of Langerhans Cells in the Lungs
Cell – Dendritic Cell

Ashley Davidoff MD
TheCommonVein.net
Langerhans Cells ? Dendritic Cells

Courtesy IMAGES IN CYTOPATHOLOGY Myunghee Kang et al Cytologic features of pulmonary langerhans cell histiocytosis in bronchial washing: A report of two cases
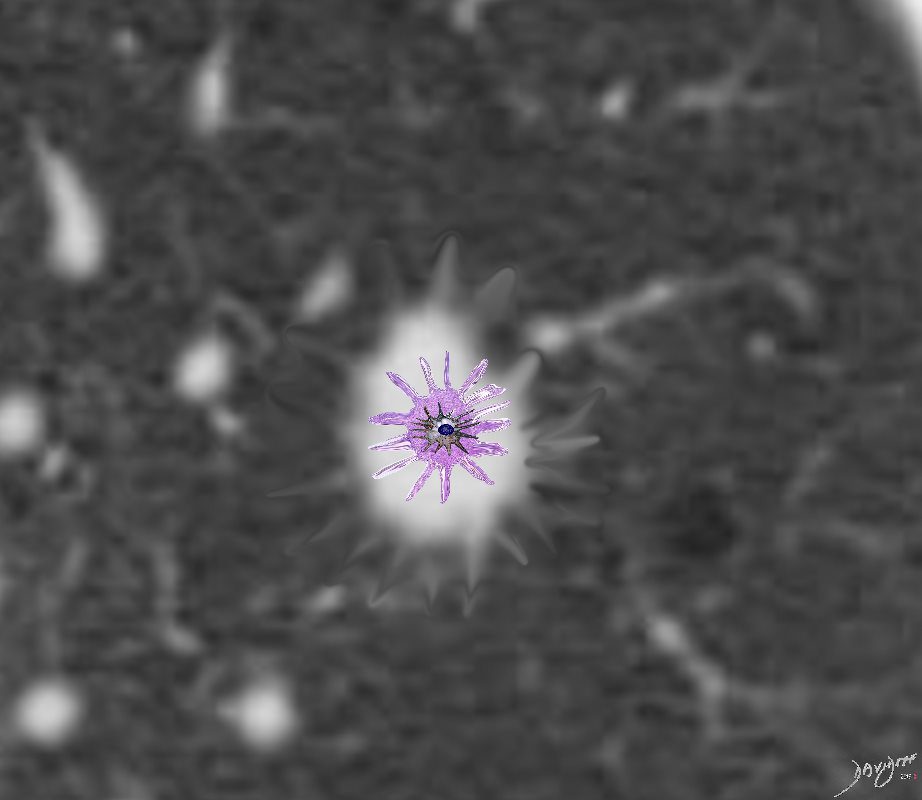
Langerhans Cell is a dendritic white cell with a wavy nucleus that creates granulomas and infiltrates the interstitium. It thus causes spiculated nodules that appear as spiculated nodules on CT
Ashley Davidoff
TheCommonVein.net

From the series “People of Israel”
Ashley Davidoff MD
TheCommonVein.net

At the level of the membranous airways the effect is predominantly related to the loss of elasticity, and aberrant accumulation of smoking related macrophages.
The weakening and destruction results in emphysema and the abnormal accumulation of smoking related macrophages relates to DIP
Ashley Davidoff
TheCommonVein.net
The Bronchocentric Nodule

Ashley Davidoff MD TheCommonVein.net
Langerhans Cells Attract Other Inflammatory cells and Surround the Bronchiole

Proliferation of the Langerhans Cell induces other cells to join causing a significant reaction around the bronchus with the inflammatory reaction extending into the interstitium and accounting for the stellate appearance
Pulmonary Langerhans Cell Histiocytosis: An Update From the Pathologists’ Perspective
March 2016 Archives of Pathology & Laboratory Medicine 140(3):230-240
Authors: Anja C Roden Eunhee S Yi
Florid Early Phase
Bronchocentric Nodules = Inflammatory Changes Around the Bronchus

Ashley Davidoff MD TheCommonVein.net lungs-0722

Ashley Davidoff MD TheCommonVein.net 50F 001b
Tree in Bud and Peribronchial Nodules with a Hint of Central Cavitation ie Bronchocentricity

Ashley Davidoff MD TheCommonVein.net lungs-0723

Ashley Davidoff MD TheCommonVein.net 50F 001

Ashley Davidoff MD TheCommonVein.net lungs-0723b
Stellate Appearance Because of Infiltration of the Inflammatory Process into the Interstitium
It may become a Solid Nodule as It Obliterates the Lumen

Ashley Davidoff MD TheCommonVein.net lungs-0724b

Ashley Davidoff MD TheCommonVein.net lungs-0724


TheCommonVein.net
Wall Starting to Break Down Creating a Thick Walled Cyst

Ashley Davidoff MD TheCommonVein.net lungs-0725
Sometimes with Bizarre Shapes

Ashley Davidoff MD TheCommonVein.net lungs-0726

Ashley Davidoff MD TheCommonVein.net 50F 001
Thick Walled Cysts = Thick Walled Dilated Small Airways

LANGERHANS HISTIOCYTOSIS
53-year-old female with nicotine dependence presents with dyspnea and cough
CXR (PA and Lateral) shows bilateral and extensive reticular nodular changes slightly more prominent in the upper lung zones
CT scan from 16 months prior showed multiple relatively thick-walled cysts predominantly in the upper lobes. The cysts are round and air filled large and are between 5mm-8mm
CT scan 9 months later shows improvement in the thickened walls of the cysts but maintenance of diffuse cystic changes predominantly in the upper lobes
A CT scan done 2 years later shows no significant change in the diffuse bilateral cystic changes, dominant in the upper lobes and consistent with Langerhans histiocytosis
Ashley Davidoff MD
With Time Inflammation Settles and There is Thinning of the Walls of the Cysts

Ashley Davidoff MD TheCommonVein.net lungs-0728b -hi res

Ashley Davidoff MD TheCommonVein.net lungs-0728b

Thin Walled Bizarre Shaped Upper Lobe Cysts
Ashley Davidoff MD TheCommonVein.net

Ashley Davidoff MD TheCommonVein.net adeno ca 007
In a Nutshell
- Unknown Etiology
- Strong association with smoking
- Isolated to lungs
- can affect multiple organs
- 20-40 years
- M.F =1:1
- Can present with PTX (10-25% spontaneous)
- Location
-
- smokers therefore
- upper mid lung involvement-
- lower lobe sparing
- costophrenic sparing
- can affect bone
- upper mid lung involvement-
- nodules
- irregular margins
- random
- decrease as disease progresses
- cysts
- thin walls
- rounded
- irregular bizarre
- progress s disease progresses
- smokers therefore
- ambivalent nature
- Can get very sick needing lung transplant
- steroid
- cytotoxic drugs
- lung transplant
- Can resolve spontaneously
- Stop smoking can resolve
- Can get very sick needing lung transplant
-
Definition
-
- Pulmonary Langerhans cell histiocytosis (PLCH)
- (previously called eosinophilic granuloma of the lung, pulmonary Langerhans cell granulomatosis, and pulmonary histiocytosis X,
- is
- a cystic interstitial lung disease
- Cause and predisposing factors
- almost exclusively in cigarette smokers.
- Caucasian young adults (20-40 years of age).
- Structures involved
- small airways terminal bronchioles and
- infiltration into the interstitium
- no to
- lymph nodes
- pleura
- Pathophysiology –
- Langerhans (immature histiocytes – myeloid dendritic cells)
- proliferate in the bronchioles and alveolar interstitium, forming granulomas
- leading to destruction of bronchioles and the interstitial alveolar space
- granulomas can cavitate
- cysts with thick walls progressing to
- cysts with thin wills which can become confluent
- Cell – Dendritic Cell

Langerhans Dendritic Macrophage
Ashley Davidoff MD
TheCommonVein.netLangerhans Cells ? Dendritic Cells

Pulmonary Langerhans cell histiocytosis.). (c) Irregular nuclear groove with abundant cytoplasm of Langerhans cells (Papanicolau stain, ×1000) (insert. CD1a).
Courtesy IMAGES IN CYTOPATHOLOGY Myunghee Kang et al Cytologic features of pulmonary langerhans cell histiocytosis in bronchial washing: A report of two cases
Figure 9. Nodular infiltrates with a stellate border extending into the surrounding interstitium in a patient with PLCH. (Courtesy of Professor A. Pesci, University of Parma.) Note
spiculated cell
can cause spiculated nodules (?due to interstitial infiltration?-
-
- Histiocytosis
- fibrous – granuloma
- small airway
- interstitial infiltrate
- Histiocytosis
-
-
- Result
- 25% can be asymptomatic and resolve spontaneously
- Clinical
- Sx
- fever, fatigue, night sweats, anorexia, weight loss) and hemoptysis
- 10% of cases, PLCH presents with
- spontaneous pneumothorax, which can be bilateral or recurrent
- Extra pulm manifestations
- bone lesions (skull, ribs, and pelvis)
- pituitary involvement producing diabetes insipidus,
- skin lesions
- Imaging
-

Spiculated Nodule of Langerhans Cell Histiocytosis
TheCommonVein.net
Langerhans Cell Histiocytosis
Langerhans Cell is a dendritic white cell with a wavy nucleus that creates granulomas and infiltrates the interstitium. It thus causes spiculated nodules that appear as spiculated nodules on CT
Ashley Davidoff
TheCommonVein.net- nodular or reticulonodular opacities
- middle and upper lung zones
- sparing of the
- bases
- costophrenic angles
- nodules
- cavitating nodules
- thick walled cysts
- thin walled bizarre shaped cysts
- preservation of lung volume
- Hilar or mediastinal adenopathy is rare
- Pleural thickening or effusion is rare
- Honeycombing in advanced disease
- FDG-PET scan
- early in the course of disease (nodular phase)
- may show increased uptake in the lungs
- PFT
- reduction in DLCO.
- total lung capacity and expiratory flow rates are well-preserved
- Restrictive, obstructive, and mixed patterns have
been described
- nodular or reticulonodular opacities
- BAL
- Bx
- transbronchial
- surgical
- Path
- Early
- cellular interstitial infiltrates of
- Langerhans? cells,
- Staining with antibodies against CD1a antigen on the cell surface
- lymphocytes,
- macrophages,
eosinophils, plasma cells, and fibroblasts
- Mid stages
- infiltrates enlarge to form nodules centered on small airways (peribronchial)
- often stellate in configuration
- Early
- Sx
- Pulmonary Langerhans cell histiocytosis (PLCH)
-
-
-
-
-
-



Spiculated Nodule of Langerhans Cell Histiocytosis in a background of Centrilobular Emphysema
TheCommonVein.net- Next Phase
- Nodules Become Cysts
- Cavitation within nodules due to
- either an
- airway remnant or
- cavitation due to an enlarging
inflammatory infiltrate
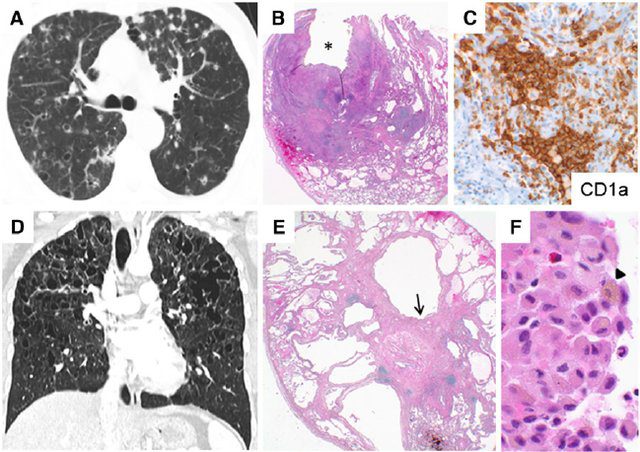
Figure 2. Pulmonary Langerhans cell histiocytosis. Multiple nodules and cysts seen on computed tomography (CT) imaging ( A ) with histology showing cellular nodules ( B ), some with central cavities ( B , *) containing diagnostic Langerhans cell aggregates highlighted by positive immunohistochemical staining for CD1a ( C , brown stain ) typical of the early cellular stage of the disease. Coronal CT image from another patient showing multiple bizarre-shaped cysts in an upper-zone?predominant distribution, with sparing of the costophrenic angles representative of later-stage disease ( D ). Histologic features typical of later disease stages include cystic spaces ( E ) associated with paucicellular stellate fibrosis ( E , arrow ). Accumulations of smoking-related pigmented macrophages ( F , arrowhead ) are frequently seen in the surrounding parenchyma. Original magnifications: 2 3 ( E ); 4 3 ( B ); 40 3 ( C ); 100 3 ( F ).
Gupta et al Diffuse Cystic Lung Disease: Part I American Journal of Respiratory and Critical Care Medicine 191(12) April 2015
Pulmonary Langerhans Cell Histiocytosis
(A) High resolution computed tomographic scan of the lung of a patient with pulmonary LCH showing the association of nodules (some of which are cavitary) and parenchymal cysts. (B) An advanced stage of the disease showing the presence of numerous cysts of various size, some of which are confluent.
Tazi, A et al Adult pulmonary Langerhans’ Cell Histiocytosis
BMJ – Thorax Volume 55, Issue 5Nodules Becoming Cysts
-

Figure 9. Nodular infiltrates with a stellate border extending into the surrounding interstitium in a patient with PLCH. (Courtesy of Professor A. Pesci, University of Parma.)
Cavitation
Cyst Formation Initially Thick Walled
Then Thin Walled Cysts
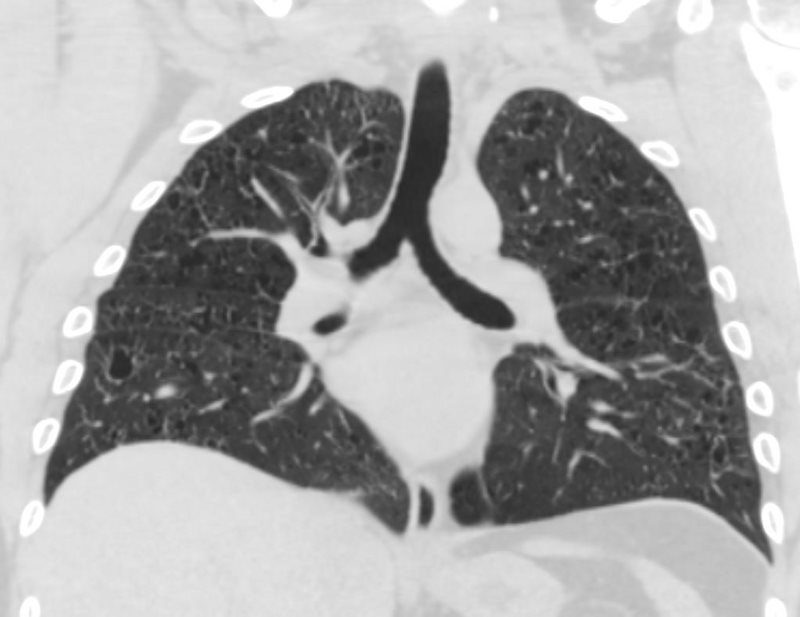
CT UPPER LUNG ZONE PROMINENCE OF SMALL THIN WALLED CYSTS
LANGERHANS HISTIOCYTOSIS
28 -year-old male with only minimal reported nicotine dependence presents with dyspnea and cough
CXR (PA and Lateral) shows no acute cardiopulmonary disease with mild interstitial prominence
CT scan showed multiple small cysts predominantly in the upper lobes. The cysts are round and air filled large and are between 5mm-8mm
These findings are consistent with Langerhans histiocytosis though the relatively minor smoking history was inconsistent with the diagnosis and thus the person was subjected to multicentric wedge biopsies.
Pathology revealed numerous stellate nodules which suggested ?burnout pulmonary Langerhans cell histiocytosis though immunostains including Langerin, CD1a and S100 were negative thus failing to confirm the diagnosis Other findings of histiocytic aggregates raised the possibility of an inhalational hypersensitivity pneumonitis.
Ashley Davidoff MD - Next Phase
- Cavitation within nodules due to
- either an airway remnant or
- de novo cavitation due to an enlarging
inflammatory infiltrate -

Figure 2. Pulmonary Langerhans cell histiocytosis. Multiple nodules and cysts seen on computed tomography (CT) imaging ( A ) with histology showing cellular nodules ( B ), some with central cavities ( B , *) containing diagnostic Langerhans cell aggregates highlighted by positive immunohistochemical staining for CD1a ( C , brown stain ) typical of the early cellular stage of the disease. Coronal CT image from another patient showing multiple bizarre-shaped cysts in an upper-zone?predominant distribution, with sparing of the costophrenic angles representative of later-stage disease ( D ). Histologic features typical of later disease stages include cystic spaces ( E ) associated with paucicellular stellate fibrosis ( E , arrow ). Accumulations of smoking-related pigmented macrophages ( F , arrowhead ) are frequently seen in the surrounding parenchyma. Original magnifications: 2 3 ( E ); 4 3 ( B ); 40 3 ( C ); 100 3 ( F ).
Gupta et al Diffuse Cystic Lung Disease: Part I American Journal of Respiratory and Critical Care Medicine 191(12) April 2015
- Costophrenic Sparing
-
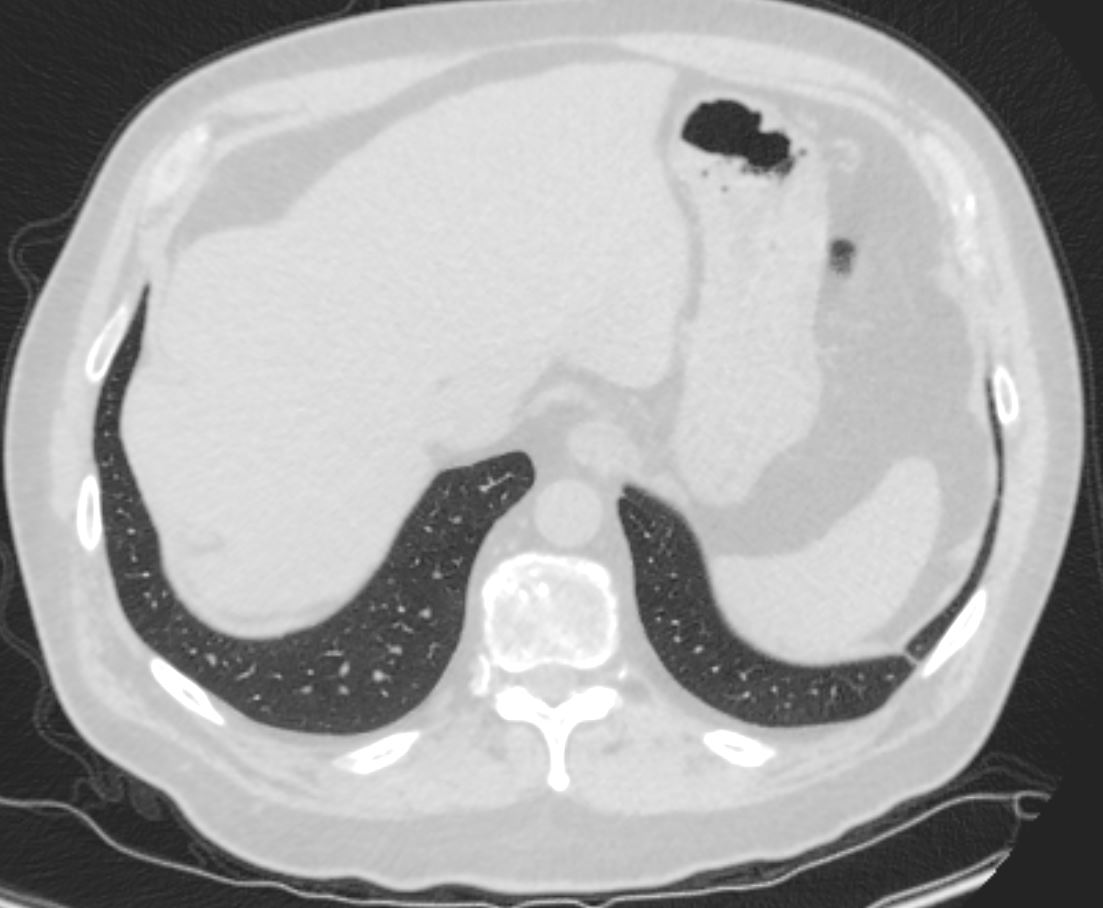
Costophrenic Sparing
Ashley Davidoff MD TheCommonVein.net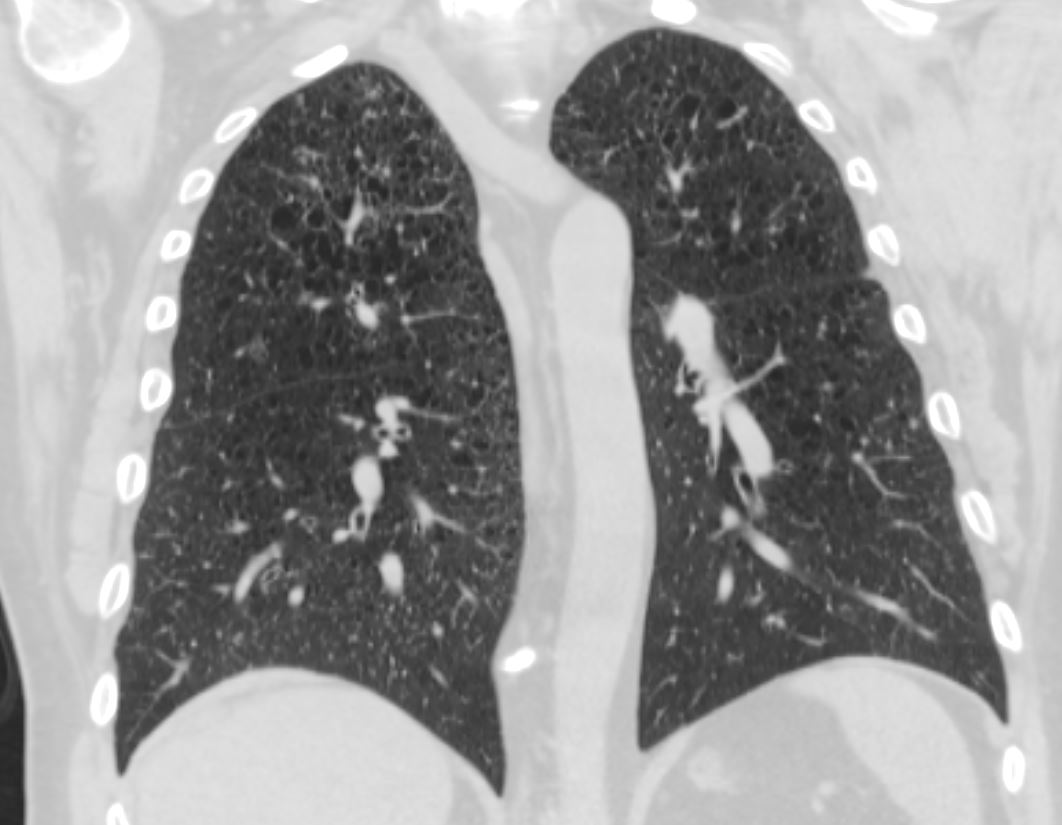
Costophrenic Sparing
Ashley Davidoff MD TheCommonVein.net66M-Langerhans-019b-CT-upper-lones-cysts.jpg
Costophrenic Sparing
Ashley Davidoff MD TheCommonVein.netSclerotic Bone Disease
66M-Langerhans-023-CTblastic-foci.jpg
Sclerotic Bone Disease possibly related to Pulmonary Langerhans Cell Histiocytosis but may just be a benign bone island
Ashley Davidoff MD TheCommonVein.net - End-stage
- prominent fibrotic scars and Langerhans cells absent
-
-
-
-
-
Clinical dyspnea or non-productive cough fatigue weight loss pleuritic pain- some asymptomatic
Some affected people recover completely after cessation of smoking,
complications such as pulmonary fibrosis and pulmonary hypertension.
Imaging
References and Links
Abbott G, et al Pulmonary Langerhans Cell Histiocytosis From the Archives of the AFIP RadioGraphicsVol. 24, No. 3 2004
Attili, A.K etal Smoking-related Interstitial Lung Disease: Radiologic-Clinical-Pathologic Correlation RadioGraphics Vol. 28, No. 5
Gupta et al Diffuse Cystic Lung Disease: Part I American Journal of Respiratory and Critical Care Medicine 191(12) April 2015
-
Videos



